Current Studies
CIU has a flexible approach to be involved in all types of clinical research and in any clinical field. A cooperation with the CIU is possible at any stage of a clinical study, even if already commenced.

CIU has a flexible approach to be involved in all types of clinical research and in any clinical field. A cooperation with the CIU is possible at any stage of a clinical study, even if already commenced.
BEready stands for "Bern, get ready" and was developed in response to the COVID-19 pandemic. The study aims to show how we can better help people in the canton of Bern to prepare for the next pandemic.

SCRIPT ("Safer Cannabis - Research In Pharmacies randomized controlled trial") is being conducted by researchers from BIHAM, DCR and ISPM in close collaboration with several other institutes at the Universities of Bern and Lucerne as well as the cities of Bern, Lucerne and Biel. The primary aim is to investigate the regulated, non-profit sale of cannabis for personal use.

The Department of The Department of Visceral Surgery and Medicine at the Bern University Hospital, University of Bern, is participating as the sole European center in an international, multicentric study funded by the National Institute of Health (NIH, USA). The primary objective of the study is to validate a novel non-invasive method for measuring portal pressure in patients with cirrhosis. The development of a precise and non-invasive technique to assess portal hypertension in this patient population would have significant implications for clinical practice.

STREAM is a randomised clinical trial supported by the Swiss National Science Foundation (SNSF).
STREAM examines the influence of statins in primary prevention in patients aged ≥70 years.
The research team consists of Prof. Nicolas Rodondi, his team at the Inselspital and the Bern Institute of Family Medicine (BIHAM), the 19 partner institutes involved throughout Switzerland and all family doctors.

“Bern, get ready” (BEready) is a so-called cohort study representing the population of the Canton of Bern to collect essential longitudinal data to improve knowledge about existing infectious diseases and preparedness for emerging health threats. BEready plans to enroll approximately 1,500 households, including adults, children, and pets, into a population-based cohort. Participants will be monitored for circulating infectious diseases.

Ciffreo is a randomized, double-blind, placebo-controlled phase III trial to evaluate the safety and efficacy of Pfizer's gene therapy construct PF-06939926 in boys with Duchenne muscular dystrophy.

A Phase 3 Multicentre, Randomized, Prospective, Open-label Trial to compare the efficacy of continuous ibrutinib monotherapy with fixed-duration venetoclax plus obinutuzumab and fixed-duration ibrutinib plus venetoclax by measuring progression-free survival (PFS) in patients with previously untreated Chronic Lymphocytic Leukaemia (CLL).
This study is investigating how Mim8 works in people with haemophilia A, who either have inhibitors or do not have inhibitors. Mim8 is a new medication that will be used for prevention of bleeding episodes. Mim8 works by replacing the function of the missing clotting factor VIII (FVIII). Mim8 will be injected with a thin needle in the skin of the stomach, using a pen-injector.
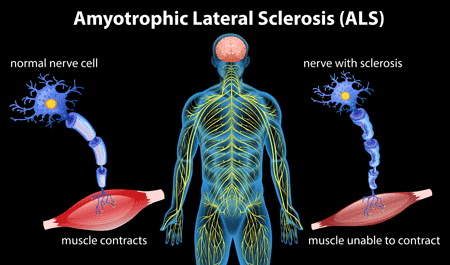
This study is being conducted to gain a better understanding of an investigational drug called edaravone as an oral/taken by mouth formulation. The oral version of edaravone is considered an investigational drug because it currently does not have regulatory approval for the treatment of ALS. The intravenous (i.v.) formulation of edaravone has been approved for the treatment of ALS in the United States, Japan, South Korea, Canada and Switzerland.

The StOP?-protocol is a short intraoperative briefing within the team present in the operating room, and it is initiated and led by the responsible surgeon. A first evaluation in a before-after study (Tschan et al., 2022) yielded reduced risks for patient mortality, length of stay, and reoperation. The current study tests if positive patient outcomes after surgical procedures using the StOP?-protocol can be confirmed in a cluster randomized trial.

Until mid-April, around 200 children and adolescents living in Switzerland, aged 6 to 17 years, will be interviewed about their diet, measured and weighed in one of the 6 study centres (Bellinzona, Bern, Lausanne, Lucerne, St. Gallen and Zurich). The pilot study aims to test the recruitment of participants and the further procedure of the survey for the main study. The start of the main study is planned for the third quarter of 2023. First results are expected in 2025
A Study of Oral Navitoclax Tablet in Combination With Oral Ruxolitinib Tablet to Assess Change in Spleen Volume in Adult Participants With Relapsed/Refractory Myelofibrosis.
A Randomized, Open-Label, Phase 3 Study Evaluating safety and change in spleen volume when navitoclax is given in combination with ruxolitinib, compared to best available therapy, for adult participants with Myelofibrosis.
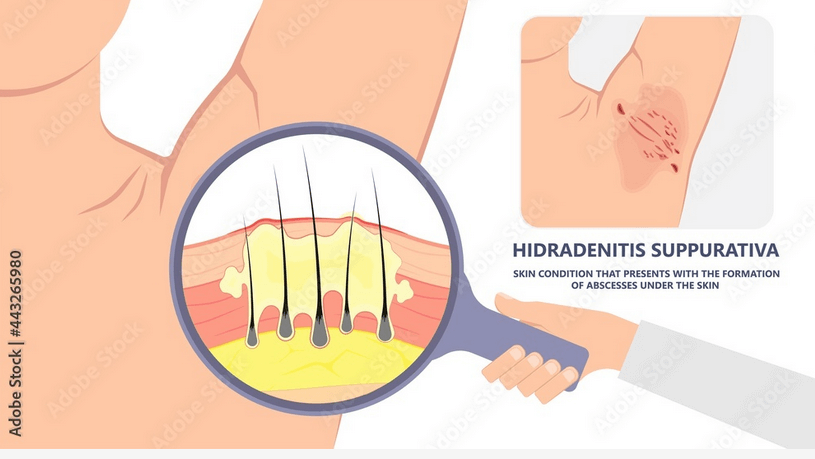
A study of Efficacy and Safety of Two Secukinumab Dose Regimens in Subjects With Moderate to Severe Hidradenitis Suppurativa (HS).
The purpose of this study is to demonstrate superiority of secukinumab at Week 16, based on Hidradenitis Suppurativa Clinical Response (HiSCR) rates versus placebo, along with the maintenance of efficacy of secukinumab at Week 52 in subjects with moderate to severe HS. Moreover, this study will also assess the safety and tolerability of secukinumab.

Prurigo nodularis is a chronic, itchy skin condition characterized by multiple raised nodules in the skin called "nodules." Doctors and researchers want to find out if nemolizumab can help people with PN.
The primary objective is to assess the effectiveness of nemolizumab (CD14152) compared with placebo after a 16-week treatment period in participants aged ≥ 18 years with prurigo nodularis (PN).
The secondary objectives are to assess the safety, pharmacokinetics (i.e., how the body processes the study treatment), and immunogenicity (i.e., what is the body's immune response to the study treatment) of nemolizumab (CD14152) compared to placebo.